已知甲醇的燃烧热数值为726.51kJ·mol-1。下列热化学方程式书写正确的是
| A.CH4O+3/2O2=CO2+2H2O △H=-726.51 kJ·mol-1 |
| B.CH4O(l)+ 3/2O2(g)=CO2(g)+2H2O(g) △H="+726.51" kJ·mol-1 |
| C.2CH4O(l)+3O2(g)=2CO2(g)+4H2O(l) △H="+1453.12" kJ·mol-1 |
| D.CH4O(l)+3/2O2(g)=CO2(g)+2H2O(l) △H=-726.51 kJ·mol-1 |
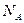 为阿伏加德罗常数)( )
为阿伏加德罗常数)( )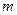 g的H2S分子含有
g的H2S分子含有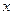 个原子,则
个原子,则 粤公网安备 44130202000953号
粤公网安备 44130202000953号