25℃、101 kPa下,碳、氢气、甲烷和葡萄糖的燃烧热依次是393.5 kJ/mol、285.8 kJ/mol、890.3 kJ/mol、2 800 kJ/mol,则下列热化学方程式正确的是
A.C(s)+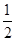 O2(g)==="CO(g)" ΔH=-393.5 kJ/mol O2(g)==="CO(g)" ΔH=-393.5 kJ/mol |
| B.2H2(g)+O2(g)===2H2O(g) ΔH=+571.6 kJ/mol |
| C.CH4(g)+2O2(g)===CO2(g)+2H2O(g) ΔH=-890.3 kJ/mol |
D.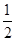 C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l) ΔH=-1 400 kJ/mol C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l) ΔH=-1 400 kJ/mol |
 粤公网安备 44130202000953号
粤公网安备 44130202000953号