下列说法正确的是
| A.已知H+(aq)+OH-(aq)===H2O(l) ΔH=-57.3 kJ·mol-1, 则H2SO4(aq)+Ba(OH)2(aq)=== BaSO4(s) +2H2O(l) ΔH=2×(-57.3) kJ·mol-1 |
| B.已知:H2(g)+F2(g)===2HF(g) ΔH=-270 kJ·mol-1,则1 mol氢气与1 mol氟气反应生成2 mol液态氟化氢放出的热量小于270 kJ |
C.500 ℃、30 MPa下,将0.5 mol N2和1.5 mol H2置于密闭的容器中充分反应生成NH3(g),放热19.3 kJ,其热化学方程式为N2(g)+3H2(g)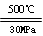 2NH3(g) ΔH=-38.6 kJ·mol-1 2NH3(g) ΔH=-38.6 kJ·mol-1 |
D.葡萄糖的燃烧热是2 800 kJ·mol-1,则 C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l) ΔH=-1 400 kJ·mol-1 C6H12O6(s)+3O2(g)===3CO2(g)+3H2O(l) ΔH=-1 400 kJ·mol-1 |
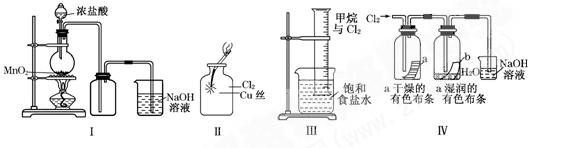
 成
成

 ①甲+H2O H2↑+乙+丙↑ ②H2 + 丙丁(气体)
①甲+H2O H2↑+乙+丙↑ ②H2 + 丙丁(气体)
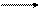 ③乙+丙甲+戊+H2O ④戊+丁甲+己(酸性)
③乙+丙甲+戊+H2O ④戊+丁甲+己(酸性) )如下表所示:
)如下表所示: 粤公网安备 44130202000953号
粤公网安备 44130202000953号