甲烷是一种高效清洁的新能源,0.25mol甲烷完全燃烧生成液态水时放出222.5kJ热量,则下列表示甲烷燃烧热的热化学方程式中正确的是
| A.2CH4(g) + 4O2(g) ="=" 2CO2(g) + 4H2O(l)ΔH=―1780 kJ·mol-1 |
| B.CH4(g) + 2O2(g) ="=" CO2(g) +2H2O(l)ΔH= +890 kJ·mol-1 |
| C.CH4(g) + 2O2(g) ="=" CO2(g) +2H2O(l)ΔH=―890 kJ·mol-1 |
| D.2CH4(g) + 4O2(g) ="=" 2CO2(g) + 4H2O(l)ΔH=+ 1780kJ·mol-1 |

 2NH3(g) ΔH=-92.4 kJ·mol-1,在反应过程中,正反应速率的变化如下图: 下列说法正确的是( )
2NH3(g) ΔH=-92.4 kJ·mol-1,在反应过程中,正反应速率的变化如下图: 下列说法正确的是( )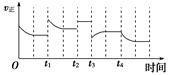
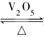 2SO3(g)是制备硫酸的重要反应。下列叙述正确的( )
2SO3(g)是制备硫酸的重要反应。下列叙述正确的( ) 一定低于反应物总能量
一定低于反应物总能量 .应用盖斯定律,
.应用盖斯定律, zC(g)。平衡时测得A
zC(g)。平衡时测得A 的浓度为0.50 mol/L,保持温度不变,将容器的容积扩大到原来的两倍,再达平衡时,测得A的浓度降低为0.30 mol/L。下列有关判断错误的是 ( )
的浓度为0.50 mol/L,保持温度不变,将容器的容积扩大到原来的两倍,再达平衡时,测得A的浓度降低为0.30 mol/L。下列有关判断错误的是 ( ) 动
动 2Z(g)一定达到化学平衡状态的是( )
2Z(g)一定达到化学平衡状态的是( ) 度不再发生变化
度不再发生变化  粤公网安备 44130202000953号
粤公网安备 44130202000953号