化学反应N2+3H2= 2NH3的能量变化如图所示,该反应的热化学方程式是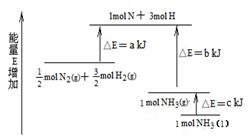
| A.N2(g)+3H2(g) = 2NH3(l)⊿H = 2(a—b—c)kJ·mol-1 |
| B.N2(g)+3H2(g) = 2NH3(g) ⊿H = 2(b—a)kJ·mol-1 |
| C.1/2N2(g)+3/2H2(g) = NH3(l) ⊿H = (b+c—a)kJ·mol-1 |
| D.1/2N2(g)+3/2H2(g) =NH3(g)⊿H = (a+b)kJ·mol-1 |
化学反应N2+3H2= 2NH3的能量变化如图所示,该反应的热化学方程式是
| A.N2(g)+3H2(g) = 2NH3(l)⊿H = 2(a—b—c)kJ·mol-1 |
| B.N2(g)+3H2(g) = 2NH3(g) ⊿H = 2(b—a)kJ·mol-1 |
| C.1/2N2(g)+3/2H2(g) = NH3(l) ⊿H = (b+c—a)kJ·mol-1 |
| D.1/2N2(g)+3/2H2(g) =NH3(g)⊿H = (a+b)kJ·mol-1 |