已知:C(s)+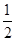 O2(g)===CO(g) ΔH=-110.5 kJ·mol-1①
O2(g)===CO(g) ΔH=-110.5 kJ·mol-1①
C(s)+O2(g)===CO2(g) ΔH=-393.51 kJ·mol-1②
计算反应C(s)+CO2(g)===2CO(g)的反应热ΔH的值为
| A.-283.01 kJ·mol-1 |
| B.+172.51 kJ·mol-1 |
| C.+283.1 kJ·mol-1 |
| D.+504.00 kJ·mol-1 |
已知:C(s)+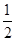 O2(g)===CO(g) ΔH=-110.5 kJ·mol-1①
O2(g)===CO(g) ΔH=-110.5 kJ·mol-1①
C(s)+O2(g)===CO2(g) ΔH=-393.51 kJ·mol-1②
计算反应C(s)+CO2(g)===2CO(g)的反应热ΔH的值为
| A.-283.01 kJ·mol-1 |
| B.+172.51 kJ·mol-1 |
| C.+283.1 kJ·mol-1 |
| D.+504.00 kJ·mol-1 |