下图是A、B、C三种物质的溶解度曲线,下列分析正确的是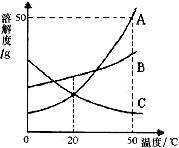
A.50℃时A、B、C三种物质的溶解度由大到小的顺序是A>B>C
B.50℃时把50gA放入50g水中能得到A的饱和溶液,其中溶质与溶液的质量比为1:2
C.将C的饱和溶液变为不饱和溶液,采用降温结晶方法
D.将50℃时A、B、C三种物质的饱和溶液降温至20℃时,这三种溶液的溶质质量分数的大小关系是B>A=C
下图是A、B、C三种物质的溶解度曲线,下列分析正确的是
A.50℃时A、B、C三种物质的溶解度由大到小的顺序是A>B>C
B.50℃时把50gA放入50g水中能得到A的饱和溶液,其中溶质与溶液的质量比为1:2
C.将C的饱和溶液变为不饱和溶液,采用降温结晶方法
D.将50℃时A、B、C三种物质的饱和溶液降温至20℃时,这三种溶液的溶质质量分数的大小关系是B>A=C