已知下列热化学方程式
Zn(s)+ 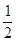 O2(g)====ZnO(s) ΔH1="-351.1" kJ·mol-1
O2(g)====ZnO(s) ΔH1="-351.1" kJ·mol-1
Hg(l)+ 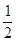 O2(g) ====HgO(s) ΔH2="-90.7" kJ·mol-1
O2(g) ====HgO(s) ΔH2="-90.7" kJ·mol-1
由此可知Zn(s)+HgO(s) ====ZnO(s)+Hg(l) ΔH3。其中ΔH3的值是( )
| A.-441.8 kJ·mol-1 | B.-254.6 kJ·mol-1 |
| C.-438.9 kJ·mol-1 | D.-260.4 kJ·mol-1 |
已知下列热化学方程式
Zn(s)+ 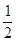 O2(g)====ZnO(s) ΔH1="-351.1" kJ·mol-1
O2(g)====ZnO(s) ΔH1="-351.1" kJ·mol-1
Hg(l)+ 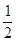 O2(g) ====HgO(s) ΔH2="-90.7" kJ·mol-1
O2(g) ====HgO(s) ΔH2="-90.7" kJ·mol-1
由此可知Zn(s)+HgO(s) ====ZnO(s)+Hg(l) ΔH3。其中ΔH3的值是( )
| A.-441.8 kJ·mol-1 | B.-254.6 kJ·mol-1 |
| C.-438.9 kJ·mol-1 | D.-260.4 kJ·mol-1 |