化学反应N2+3H2=2NH3的能量变化如图所示,E是正值,该反应的热化学方程式是( )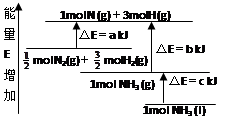
| A.N2(g)+3H2(g)=2NH3(1); △H=2(a-b-c)kJ·mol-1 |
| B.N2(g)+3H2(g)=2NH3(g);△H=2(b-a)kJ·mol-1 |
C. N2(g)+ N2(g)+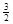 H2(g)=NH3(1);△H=(b+c-a)kJ·mol-1 H2(g)=NH3(1);△H=(b+c-a)kJ·mol-1 |
D. N2(g)+ N2(g)+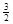 H2(g)=NH3(g); △H=(a+b)kJ·mol H2(g)=NH3(g); △H=(a+b)kJ·mol |
化学反应N2+3H2=2NH3的能量变化如图所示,E是正值,该反应的热化学方程式是( )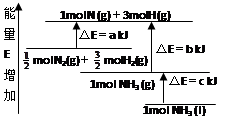
| A.N2(g)+3H2(g)=2NH3(1); △H=2(a-b-c)kJ·mol-1 |
| B.N2(g)+3H2(g)=2NH3(g);△H=2(b-a)kJ·mol-1 |
C. N2(g)+ N2(g)+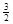 H2(g)=NH3(1);△H=(b+c-a)kJ·mol-1 H2(g)=NH3(1);△H=(b+c-a)kJ·mol-1 |
D. N2(g)+ N2(g)+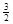 H2(g)=NH3(g); △H=(a+b)kJ·mol H2(g)=NH3(g); △H=(a+b)kJ·mol |