发射卫星用N2H4为燃料,NO2为氧化剂,两者反应生成N2和水蒸气, 已知
N2(g)+2O2(g)=2NO2(g) ⊿ H1 = + 67.7kJ/mol;
N2H4(g)+O2(g)= N2(g)+2H2O(g) ⊿ H2 = - 534kJ/mol;
2H2(g)+ O2(g)= 2H2O(g) ⊿ H3 = - 484kJ/mol;
H2(g)+F2(g)=2HF(g) ⊿ H4 = - 538kJ/mol
写出N2H4与 NO2反应的热化学方程式________________________________________,
写出N2H4与F2反应的热化学方程式__________________________________________。
 2C(g);△H<0,平衡常数为K。其它条件不变时,温度升高,平衡常数K
2C(g);△H<0,平衡常数为K。其它条件不变时,温度升高,平衡常数K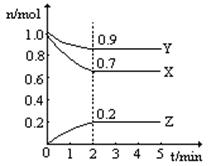
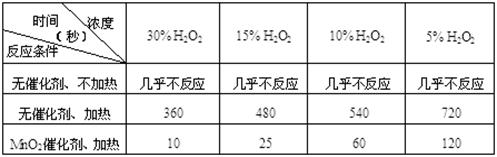
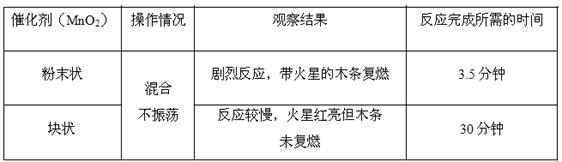
 粤公网安备 44130202000953号
粤公网安备 44130202000953号