已知:101kPa时,辛烷的燃烧热为5518kJ·mol-1,强酸与强碱在稀溶液中发生反应时的中和热为57.3kJ·mol-1,则下列热化学方程式书写正确的是 ( )
① C8H18(l)+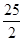 O2(g)= 8CO2(g)+9H2O(g);△H = -5518kJ·mol-1
O2(g)= 8CO2(g)+9H2O(g);△H = -5518kJ·mol-1
② C8H18(l)+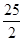 O2(g)= 8CO2(g)+9H2O(l);△H = -5518kJ·mol-1
O2(g)= 8CO2(g)+9H2O(l);△H = -5518kJ·mol-1
③ H+ + OH- =" " H2O;△H = -57.3kJ·mol-1
④ NaOH(aq)+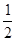 H2SO4(aq)
H2SO4(aq)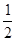 Na2SO4(aq)+H2O(l);△H= +57.3kJ·mol-1
Na2SO4(aq)+H2O(l);△H= +57.3kJ·mol-1
| A.①③ | B.② | C.②④ | D.②③ |

 粤公网安备 44130202000953号
粤公网安备 44130202000953号