已知下列反应的热化学方程式为:C(s)+O2(g)=CO2(g) △H1=—393.5kJ/niol
CH3COOH(1)+2O2(g)= 2CO2(g)+ 2H2O(1) △H2="-" 870.3kJ/mol
H2(g)+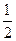 O2(g)=H2O(1) △H="-285.8" kJ/mol
O2(g)=H2O(1) △H="-285.8" kJ/mol
则2C(s)+2H2(g)+O2(g)=CH2COOH(1)的反应热△H为
| A.+488.3kJ/mol | B.-488.3kJ/mol |
| C.-244.15kJ/mol | D.+244.15kJ/mol |
已知下列反应的热化学方程式为:C(s)+O2(g)=CO2(g) △H1=—393.5kJ/niol
CH3COOH(1)+2O2(g)= 2CO2(g)+ 2H2O(1) △H2="-" 870.3kJ/mol
H2(g)+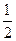 O2(g)=H2O(1) △H="-285.8" kJ/mol
O2(g)=H2O(1) △H="-285.8" kJ/mol
则2C(s)+2H2(g)+O2(g)=CH2COOH(1)的反应热△H为
| A.+488.3kJ/mol | B.-488.3kJ/mol |
| C.-244.15kJ/mol | D.+244.15kJ/mol |